|
Laparoscopic Subtotal Gastrectomy for Gastric Cancer. JSLS. 2. 00. 9 Jul- Sep; 1. Danny Rosin, MD,Yuri Goldes, MD, Barak Bar Zakai, MD, Moshe Shabtai, MD, Amram Ayalon, MD, and Oded Zmora, MDThe Department of General Surgery and Transplantation, Sheba Medical Center, Tel Hashomer, Sackler School of Medicine, Tel Aviv University, Israel. Corresponding author. Address reprint requests to: Danny Rosin, MD, The Department of General Surgery and Transplantation, Sheba Medical Center, Tel Hashomer, Israel. 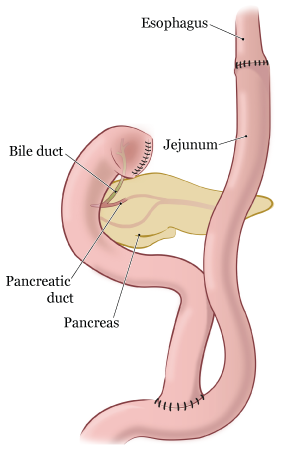
Post-Gastrectomy and Anti-Dumping Diet For those who have lost part of their stomach due to surgery. Gastrectomy Nutrition Guidelines. How should I eat after a gastrectomy to prevent dumping syndrome? A gastrectomy is a partial or total surgical removal of the stomach. Gastrectomies are performed to treat stomach cancer and perforations of the. After your gastrectomy or esophagogastrectomy. If you have questions about your diet while you are in the hospital, ask to see your unit dietitian. Subtotal gastrectomy for stomach cancer. A subtotal gastrectomy: Is an inpatient procedure. Typically patients stay in the hospital for about a week.    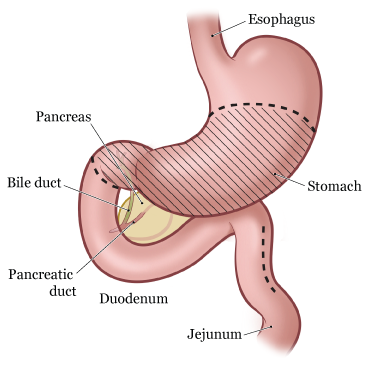 
Telephone: 9. 72–5. Fax: 9. 72–3- 5. 30. E- mail: moc. cam@nisord. Copyright . However, several reports suggest that the laparoscopic approach may be safe and applicable. The aim of this study was to review our experience with laparoscopic gastrectomy for gastric malignant tumors amenable to subtotal gastrectomy, and assess the oncologic outcome. Methods: The laparoscopic approach to subtotal gastrectomy was selected according to both the surgeon's and patient's preference. Data regarding demographics, operative procedures, postoperative course, and follow- up were prospectively collected in a computerized database. Survival data were obtained from the national census. Results: Twenty patients were operated on, 1. There were 1. 0 males and 1. D1 subtotal gastrectomy with Billroth- 2 reconstruction was performed. Mean operative time was 3. Tumor- free margins were obtained in all cases, and a mean of 1. Median postoperative hospital stay was 1. Postoperative complications included leak from the duodenal stump (2), intraabdominal abscess (2), anastomotic leak (1), wound infection (1), and bowel obstruction (1); reoperation was required in 4 patients. No perioperative mortality occurred in our series. Pathology showed nodal involvement in 8 patients. During a mean follow- up of 3. The Kaplan- Meier calculated 5- year survival was 7. Conclusion: Although a challenging and lengthy procedure, laparoscopic subtotal gastrectomy yields acceptable surgical and oncologic results that may further improve with increased surgeon experience. Thus, the application of laparoscopy in the surgical treatment of distal gastric malignancy may be considered; however, further data are needed before this approach can be recommended. Keywords: Laparoscopy, Gastrectomy, Malignancy. INTRODUCTIONDespite the rapid development and application of laparoscopy for a wide variety of pathologies, 2 major factors continue to limit its widespread use for gastric malignancy: technical complexity requiring advanced laparoscopic skills and concerns regarding oncologic safety. Specifically, the laparoscopic approach involves a lengthier and more complex procedure with the need for specialized equipment and a high level of dexterity. Thus, many surgeons opt for the traditional open technique, especially when dealing with malignancy, where concerns that the laparoscopic approach may not yield similar results to those achieved by open surgery, such as the extent of resection and recurrence. To adopt a radical change for the preference of laparoscopy over open surgery for gastric malignancy, justification must first be presented by way of a demonstrated significant advantage. For these reasons, laparoscopic gastric resection is not currently widely practiced, and as a result data relative to its safety and efficacy are limited. Another limiting factor in reaching any evidence- based conclusions regarding laparoscopy for gastric malignancy is the relatively low incidence of this type of cancer in the Western hemisphere. Conversely, colonic cancer has a much wider prevalence that has allowed for a plethora of data establishing the safety of laparoscopic colectomy. In Japan, for example, where the incidence of gastric cancer is higher, laparoscopic gastrectomy has been performed for more than 1. However, it may be difficult to extrapolate from the Japanese experience, because more stringent population screening leads to earlier detection. In addition, even when diagnosed at an operable stage, the overall prognosis of gastric cancer is more dismal in Western countries than in Japan. Although reported results are limited, based on the promising experiences of laparoscopic gastrectomy from several European and North American Centers, in 2. The aim of this study was to review our experience with laparoscopic subtotal gastrectomy for gastric malignancy, focusing specifically on the perioperative course and postoperative oncologic results. METHODSPatient Selection. All patients diagnosed with resectable gastric malignancy at the distal stomach between 2. Patients were selected for the laparoscopic approach based on both the surgeon's and patient's preference. Tumor size was not considered as an exclusion criterion. The final decision regarding the selected surgical approach was made by the operating surgeon. The choice of subtotal gastrectomy was based on the location of the tumor in the stomach, which allowed sufficient resection margins with ample residual proximal stomach for the creation of the gastrojejunal anastomosis. Data Collection and Analysis. Data regarding patient demographics, type of surgery, intraoperative and postoperative course, including complications and repeat procedures, pathologic results, adjuvant and neoadjuvant therapy, and long- term follow- up, including late complications, local recurrence, metastatic spread, and survival were prospectively entered into a computerized database and retrospectively analyzed. Early postoperative complications were defined as those occurring immediately following surgery or within 2 weeks after the procedure. Follow- up data were obtained from the patient's routine outpatient clinic visits. National census data were used to determine survival time. Operative Technique. Surgery was performed with the patient in the supine position and the surgeon standing between the patient's legs. Abdominal entry was made immediately above the umbilicus, by the open Hasson technique (for patients who had prior surgery) or a Veress needle, for initial CO2 insufflation; 4 additional ports were usually necessary to complete the procedure. Abdominal exploration was initially performed to rule out metastatic spread. The tumor location at the stomach was confirmed visually and by instrumental palpation, to determine the appropriate surgical procedure. Resection commenced with mobilization of the omentum from its attachments to the transverse colon, for inclusion in the surgical specimen. This dissection allows entry into the lesser sac to examine the posterior gastric wall and rule out posterior tumoral penetration into the pancreas (Figure 1). Further dissection was carried out distally, along the greater curve of the stomach, towards the gastric outlet; care was taken to avoid the transverse mesocolon while transecting the right gastroepiploic artery. The first part of the duodenum was then dissected and transected using a linear stapler; sutures were usually added to reinforce the duodenal closure. Proximal dissection along the greater curvature was then carried out to the proximal tumor margins, at least 5cm from the tumor. The short gastric vessels to the spleen were then transected to a height depending on the level of transection. At this stage, dissection was continued at the lesser curvature side of the stomach, with division of the right gastric artery near the antrum. The left gastric artery is usually divided at its origin if high transection is required or at its branches if more proximal stomach is to be retained. Generally, perigastric lymph nodes were removed with the specimen (D1 dissection), and no attempt was made to dissect distant nodes (D2 dissection), unless evidently enlarged (similar to our open approach). Lesser sac entry and examining posterior gastric wall and pancreas. After dissection was completed, the stomach was horizontally transected using several cartridges of a linear cutting stapler (Figure 2). The specimen was removed in a collection bag through a 4- cm to 5- cm upper abdominal incision. Frozen section pathologic examination of the surgical margins was carried out to ensure tumor- free margins before proceeding with the anastomosis. Gastric transection using a linear stapler. Gastrointestinal tract continuity was restored in a “Billroth 2” configuration, whereby a jejunal loop was anastomosed to the gastric stump. The bowel was passed anterior to the transverse colon, and anastomosed to the posterior gastric wall using a 6. RESULTSOf nearly 4. Indications for surgery included adenocarcinoma in 1. GIST) in another. All tumors were located in the distal part of the stomach and were amenable to subtotal gastrectomy. No patients required neoadjuvant oncologic treatment. Adjuvant treatment was offered to patients at a high risk of experiencing a recurrence (Stage above T2. N0, poorly differentiated tumor). All procedures were completed laparoscopically (no conversion to open surgery were needed). In 2 patients, intraoperative endoscopy was required to assist in locating a tumor that was not visible or palpable using the laparoscopic equipment. The mean operating time was 3. An intraoperative complication occurred in only one patient, wherein the esophageal thermometer was mistakenly placed in the stomach and subsequently included in the staple line, necessitating extension of the resection and refiring of the stapler. Nine early postoperative complications occurred in 6 (3. Table 1). Duodenal stump leak, occurring in 2 patients in whom the staple line was not reinforced, was conservatively treated in one patient but required reoperation in the other. Two patients developed an intraabdominal abscess; one was drained percutaneously, and the other was drained by repeat laparoscopy. Leak from the gastrojejunal anastomosis led to reexploration in one patient (who also had a duodenal stump leak), revealing ischemia of the proximal stomach. Extension of the gastric resection with reanastomosis to the small bowel led to eventual recovery.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
May 2017
Categories |
 RSS Feed
RSS Feed
